11 Tests for Face Mask on-site Inspection in China
As a face mask is directly in contact with skin for potentially long periods of time, relevant restricted substance and biocompatibility testing are highly encouraged. Also, the ear loops have to be securely attached to a reusable face mask for the duration of its expected lifetime.

We draw samples from production and evaluate them according to the agreed test protocol. We will also verify that they meet the applicable regulatory requirements, through document review or actual mask testing.
TESTCOO guided by the relevant Standards, except for processing visual inspection - a visual and physical examination of labelling, design and construction quality, is also testing the following attributes of face masks:
- Barcode scan check: The barcode must be scannable and with the correct number.
- Product Size /Weight measurement: According to product spec or approved sample. Apply +/- 3% if no detail tolerance issued or requirement from client.
- Fitting check: The product should be fit the intended model or person well.
- Odor and smell check: Should not have irritative smell and odor.
- Tension test for ear loop: Test with 20N static tensile strength for 5s, the connection point between each mask belt and body shall not be broken.
- Mask internal check: Open the mask to check inside fabric. No contamination or foreign fiber, not contain any hard and sharp contaminants.
- Combustion test for mask: The mask should be made of non- flammable materials. The combustion of the mask should be no more than 5s after leaving the flame.
- Color fastness test: By using a wet white cloth, rub on the inspected cloth back and forth 5 times, to see if there is any color left on the white cloth.
- Polarity check -Prepare some paper scraps smaller than 1x1cm: Fully contact the melt-blown material with paper scraps; Gently lift the melt- blown material and observe the adsorption effect of paper scraps - -The paper scraps should obvious polar adsorption effect on melt-blown materials
- Filtration efficiency test: Actual Finding should match the requirement/tolerance.
- GSM test per fabric type (not applicable for sweater/cardigan or any other woolen product: According to client's specification. Apply +/- 3% tolerance if no detail tolerance issued or requirement from client.
As the recognized global benchmark for quality and integrity, we will help you to ensure that your products meet government and consumer demands for quality and safety.
More Facemasks
-------------------------------------------------------------------------------------------------------------
Utility mask
A simple, flexible barrier that is placed over the nose and mouth during short exams and visitations, or during short procedures that do not produce fluid, spray or aerosols. Utility masks are not regulated as medical devices and are not recommended to provide protection from COVID-19.

Surgical mask
A fluid-resistant, disposable device intended to be placed over the nose and mouth of medical personnel or patients who are infected or displaying symptoms, to create a physical barrier between the mouth and nose of the wearer and prevent the transmission of airborne organisms during surgery or patient examination. Surgical masks are graded as level 1, 2 or 3 based on the level of protection provided, or fluid resistance.

Respirators
A filtering face piece respirator (FFR) that is designed to form a very close seal around the nose and mouth, protecting the wearer from exposure to airborne particles including pathogenic biological airborne particulates such as viruses and bacteria. Respirators are often labelled with the effective particulate filtration they have been tested for to ensure they remove the minimum specified percentage of solid and liquid aerosols. For example, N95 respirators are effective in removing a minimum of 95% of solid and liquid aerosols that do not contain oil. Respirators are a single use item.
In addition to the N95 designation (with reference to the United States NIOSH 42 CFR Part 84), there are other FFR classes including FFP2 (Europe EN 149-2001), KN95 (China, GB2626-2019), P2 (Australia/ New Zealand AS/NZS 1716:2012). A comparison of these standards are provided at: Comparison of P2, FFP2, KN95, and N95 and Other Filtering Facepiece Respirator Classes(link is external)
The consumer or user of respirators should review the similarities and differences between respirators in terms of their specifications and consider their suitability for the settings in which they will be used.

Surgical respirator
A filtering face piece respirator with a similar structure and design to standard respirators to protect the wearer from exposure to pathogenic biological airborne particulates, therefore meeting the same testing requirements for a minimum percentage of filtration against airborne particulates.
Surgical respirators have also been tested for fluid resistance against penetration by synthetic blood under different pressures, such as may occur during certain high risk medical procedures.
Note
The TESTCOO is prioritising and expediting all COVID-19-related assessments, reviews, and responses to queries.
For more information about face masks and get a face mask free sample report, contact us at service@testcoo.com.
Free Sample Report Performance Quality Control
Download a sample report to keep control of your supply chain!
Featured Articles
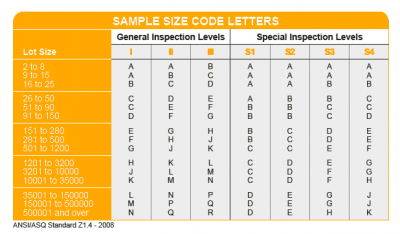 AQL Table | How to Read It
AQL Table | How to Read It TOP 10 Common Defects in Garments Quality Inspection
TOP 10 Common Defects in Garments Quality Inspection Product Packaging and Shipment Label requirements for Amazon FBA
Product Packaging and Shipment Label requirements for Amazon FBA What Is ASTM-F2413-18? Protective Footwear Standard
What Is ASTM-F2413-18? Protective Footwear Standard How to Conduct Third-Party Quality Control Inspections for Electric Scooters
How to Conduct Third-Party Quality Control Inspections for Electric Scooters SMETA Audit-What is SMETA Audit?
SMETA Audit-What is SMETA Audit? TESTCOO Supplier Verification/Certification Service SLCP, Higg FEM, GRS, GOTS
TESTCOO Supplier Verification/Certification Service SLCP, Higg FEM, GRS, GOTS Quality Control Inspection Company in China
Quality Control Inspection Company in China What is Quality Inspection? A Complete Guide
What is Quality Inspection? A Complete Guide Guidelines for Product Inspection in India
Guidelines for Product Inspection in India
Category
- Production Inspection Service
- Factory Audit
- Softline Inspection
- Hardline Inspection
- Electrics Inspection
- Certification
- Checklist
- Manufacturers
- Quality Assurance Basics
- Products Recall
- AQL
- Guidence and Standard
- News
- Supplier Management
- Amazon
- Protective Equipment
- e-commerce quality control
- Indian Manufacturing
- Soft Goods Quality Control
- Supply Chain Management
- Supply Chain Resilience
- E-Commerce Quality Control
- ISO 2859
- Supply Chain Optimization
- Garment Industry
- Higg Index



